Feed mills should have a plan for minimizing the risk of pathogens.
July 24, 2018
By Wilmer Pacheco, Adam Fahrenholz and Charles Stark*
*Dr. Wilmer Pacheco is an extension specialist and assistant professor in the department of poultry science at Auburn University. Dr. Adam Fahrenholz is assistant professor in the Prestage department of poultry science at North Carolina State University. Dr. Charles Stark is the Jim & Carol Brown associate professor in feed technology at Kansas State University.
Minimizing the risk of pathogens in a feed mill usually becomes a hot topic when a problem arises in the field. However, feed mills should have a plan for minimizing the risk of pathogens.
In this month’s article let’s look at the different options currently available to the feed industry to minimize the risk of pathogens in the feed mill.
In many cases, a hazard analysis may in fact lead a facility to conclude that pathogens are not a significant concern in their specific situation. However, the question of pathogen control, both the need for and how to accomplish it, is often raised. So, for the purpose of this article, let’s assume we have a facility that has decided to make a particular effort to minimize pathogens in animal feeds.
Producing a guaranteed pathogen-free animal feed may be difficult because we operate in a dynamic system where pathogens, which are invisible to the naked eye, can survive and be transferred throughout the feed mill and potentially to animal feeds. Pathogens can enter through feed ingredients, the environment, deficiencies in process controls and facility design, as well as through poor employee practices due to a lack of proper training.
Ingredient suppliers and animal food producers have made good progress in reducing salmonella prevalence in feed ingredients and animals’ foods; for example between the years 2002 and 2006, 30.9% of ingredient samples analyzed were contaminated with salmonella; however, only 19.4% were contaminated from 2007 to 2009. This represented a reduction of around 40% (Li et al., 2012). Nevertheless, a feed mill may also wish to pursue additional strategies to reduce pathogens and prevent recontamination during feed manufacturing, transportation, and storage.
At the feed mill, improved microbial control can be accomplished by following Current Good Manufacturing Practices (CGMPs), other facility-specific prerequisite programs such as employee training, cleaning/sanitation and quality assurance as well as potentially through preventive controls such as supply chain applied controls, process controls and sanitation controls. In addition, dust control to prevent contamination by environmental pathogens as well as humans and vehicle traffic control to prevent cross-contamination should be part of an integrated program to minimize pathogens in animal feeds.
Each feed mill, regardless of the purpose (commercial or integrated), should create a team to work together to identify, evaluate, and control hazards associated with their facility or type of animal feeds they are producing. Ingredient selection and quality control during receiving are the first lines of defense to produce pathogen-free animal feeds. As the pathogen load increases in feed ingredients, it not only becomes more difficult to eliminate, but it also increases the risk of cross-contamination at the feed mill. Therefore, the quality assurance team should focus significant efforts towards having good ingredient specifications, buying ingredients only from approved and trusted suppliers, and possibly verifying production and transportation procedures through periodic visits to the supplier. In addition, the quality assurance program should provide procedures that scrutinize and analyze deliveries and reject ingredients that don’t meet the standards previously defined.
Housekeeping also serves an important role as pathogens can be transferred through dust, and poor housekeeping can therefore increase the risk of cross-contamination. In order to perform good housekeeping activities, utensils or tools such as brooms, shovels, etc., should always be in good working condition and they should be separated by working areas such as ingredient receiving, processing and load out. A routine cleaning and sanitation schedule for these tools will help minimize the risk of spreading pathogens throughout the feed mill. Using color-coding for cleaning tools may be necessary to prevent their movement from a dirty to a clean area. Pathogens can also be transferred by personnel; therefore, the usage of uniforms with different colors depending on the area, can also prevent cross-contamination. Additionally, adding dry or wet foot baths at entry points to the feed mill will help minimize the risk of pathogens. However, these foot baths must be routinely monitored to prevent them from becoming a source of pathogens.
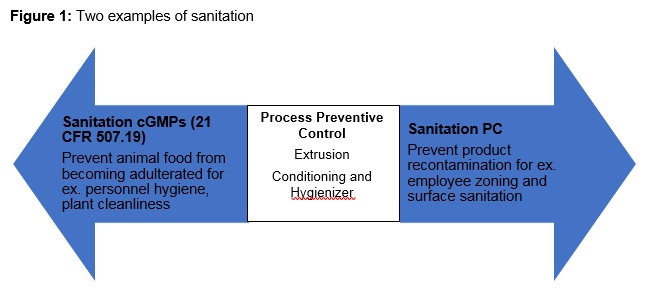
In the event that a preventive control is deemed necessary, two types of sanitation should be employed: sanitation CGMP’s before the process preventive control of the pathogen, and sanitation controls after the process preventive control (Figure 1). The most effective step to control pathogens in feed mills is through heating and moisture addition during the conditioning process. For example, companies focused on producing Salmonella-free feeds are relying on conditioning their feeds to at least 85°C and retaining it using a hygienizer for more than three minutes. In simple terms, a hygienizer is a tube with steam jacketing where feed is moved through by a screw auger using the first in, first out (FIFO) principle. After the hygienizer, the feed enters into a standard pellet mill where mash is agglomerated and shaped between a pellet die and two or three rolls, depending on the design. Additional research is necessary to optimize the heating process in order to kill salmonella without deactivating heat labile nutrients such as vitamins and amino acids.
Chemical compounds such as formaldehyde and organic acids are becoming more popular to prevent recontamination after the killing step. Formaldehyde has been widely used as a biocide broadly effective in destroying a wide range of organisms and is used to prevent salmonella outbreaks due to contamination in poultry and pig feeds. However, it can interact with lysine and render it unavailable, at least partially. Furthermore, in December 2017, at the Standing Committee on Plants, Animals, Food & Feed (SCOPAFF) meeting, 26 nations of the European Union voted against the usage of formaldehyde as a feed additive. Therefore, feed mills in the European Union will no longer be able to include formaldehyde to prevent contamination after the heat treatment step. Organic acids such as propionic and formic acids are often used to treat grains in silos and prevent mold growth and bacterial re-contamination after heat treatment. The addition of these chemical compounds prior to pelleting appears to offer better protection against recontamination, as post-pellet application only offers protection on the surface of the pellet.
Regardless of the process step to kill salmonella and the usage of chemical compounds to prevent recontamination, the pellet cooler would need to be fully enclosed and receive clean filtered air derived from an area of the feed mill that is dust free in order to most effectively prevent re-contamination with airborne bacteria. In addition, the cooler should be managed to reduce the amount of moisture in the feed to less than 13% to prevent bacterial and fungal growth. Verification procedures such as feed sampling and environmental monitoring for environmental pathogens are important to ensure that salmonella-free feeds is produced.
In the most pathogen-sensitive facilities, load out areas could be separated from the manufacturing areas and could be pressurized and be equipped with doors that automatically close on truck entry and during the feed loading process. In addition, trucks can be heated before entering the loading area to prevent cross-contamination. Good truck sanitation is essential as poorly cleaned trucks can lead to re-contamination with pathogens. In addition, to ensure that the animals are truly receiving pathogen-free feeds, bins at the farm must always be cleaned and sealed to prevent re-contamination. The production of pathogen-free feeds should start with effective ingredient testing programs, sanitation CGMPs (21 CFR 597.19), effective process controls to deactivate the pathogen of interest, sanitation preventive controls, effective microbial testing programs, pest elimination (see last month’s article), dust and air controls, moisture minimization, transportation in clean and dry systems and control of re-contamination at the farm. Feed sanitation is an integrated process and the weakest link in the biosecurity process will generate contaminated animal feeds.
References
Li X., L. A. Bethune, Y. Jia, R. A. Lovell, T. A. Proescholdt, S. A. Benz, T. C. Schell, G. Kaplan, D. G. McChesney. 2012. Surveillance of Salmonella prevalence in animal feeds and characterization of the Salmonella isolates by serotyping and antimicrobial susceptibility. Foodborne Pathog Dis 9(8):692-8.
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
